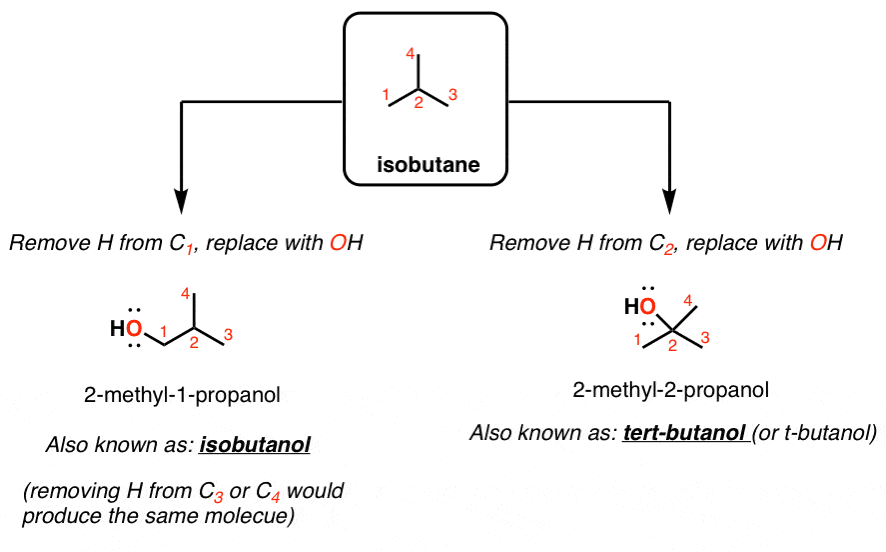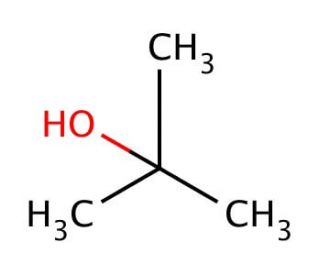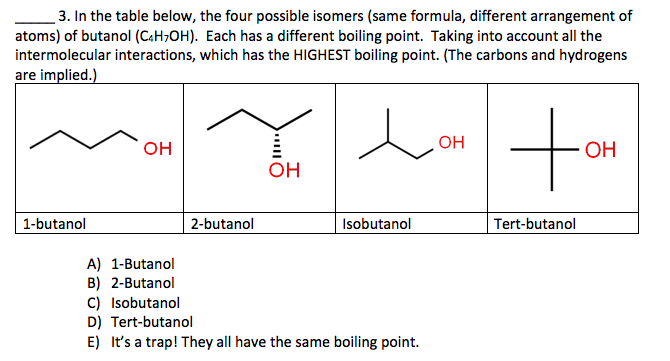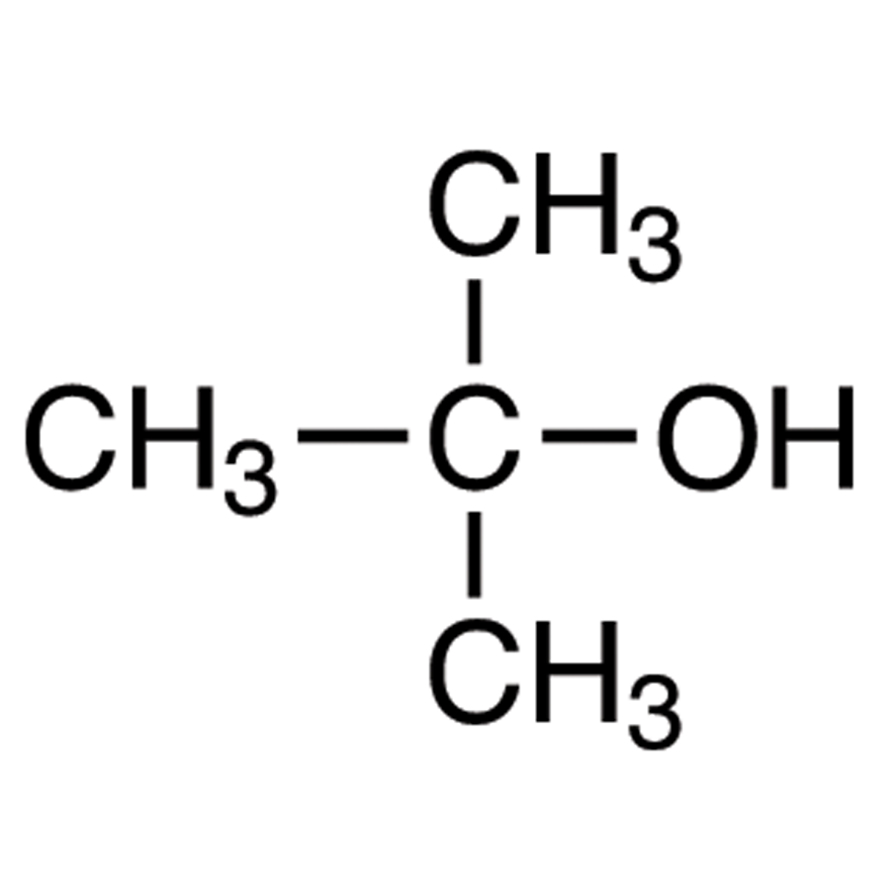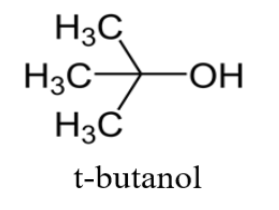
Correct statement(s) in case of n-butanol and t-butanol is (are):A. both are having equal solubility in water.B. t-butanol is more soluble in water than n-butanol.C. The boiling point of t-butanol is lower

Impact of a tert-butyl alcohol-cyclohexane system used in unidirectional freeze-casting of SiOC on compressive strength and mass transport - ScienceDirect

Why are the boiling points for butanol, isobutanol, and t-butyl alcohol different? | Homework.Study.com